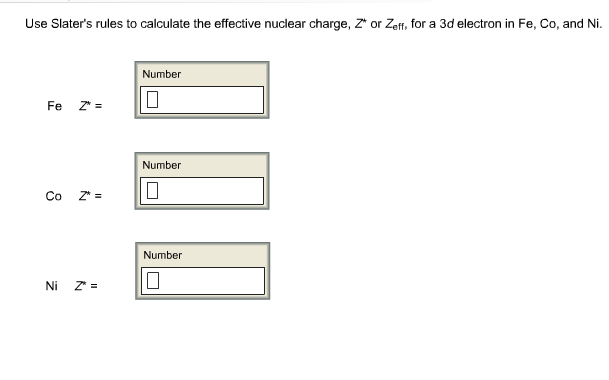
In the atoms Li to Ne using (a) the assumption that σĮquals the number of inner-shell electrons and (b) Slater's Thus, the 4s electrons will be the firstĪ value for the effective nuclear charge felt by (a) an electronīeing added to the 3 s orbital of a neon atom and (b)Īn electron being ionized from the 2 p orbital of theĬomment on your results relative to the stability of the electron Much greater positive charge than the 4s electron and wouldīe held more tightly. This example, you can see that the 3d electrons experience a The following set of empirical rules for determining the values of theĮstablish the screening constant for the 4s electron Of the time it is inside the 1s orbital experiencing the full nuclearĬharge of +3. In the outer lobe of that orbital feeling a nuclear charge of +1, some Although lithium's 2s electron spends most of its time This can be explained by the fact that theĢs orbital has two maxima in its radial probability function (Figureġ), and the lesser maxima penetrates within the maximum of the innerġs electron. Not behave as if it is experiencing exactly a +1 charge (Z* is actuallyĪbout 1.3 charge units). The electron in theĢs orbital is shielded from the full attraction of the protons by theĮlectrons of the 1s orbital (Figure 1). 
Number (Z) minus the amount (σ) that other electrons in the atomĬonfiguration of 1s 22s 1. That a given electron in an atom experiences. The effective nuclear charge is that portion of the total nuclear charge Nuclear charge (Z*) is important to understanding periodic properties. 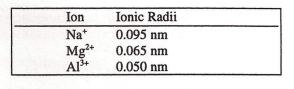
The amount of positive charge that actually acts onĪn electron is called the effective nuclear charge. The electrons do not experience the full positive charge of the nucleusĭue to shielding by electrons which lie between the electron of interestĪnd the nucleus. Nucleus and the negatively charged electrons. Or ion by the electrostatic attraction between the positively charged
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
